






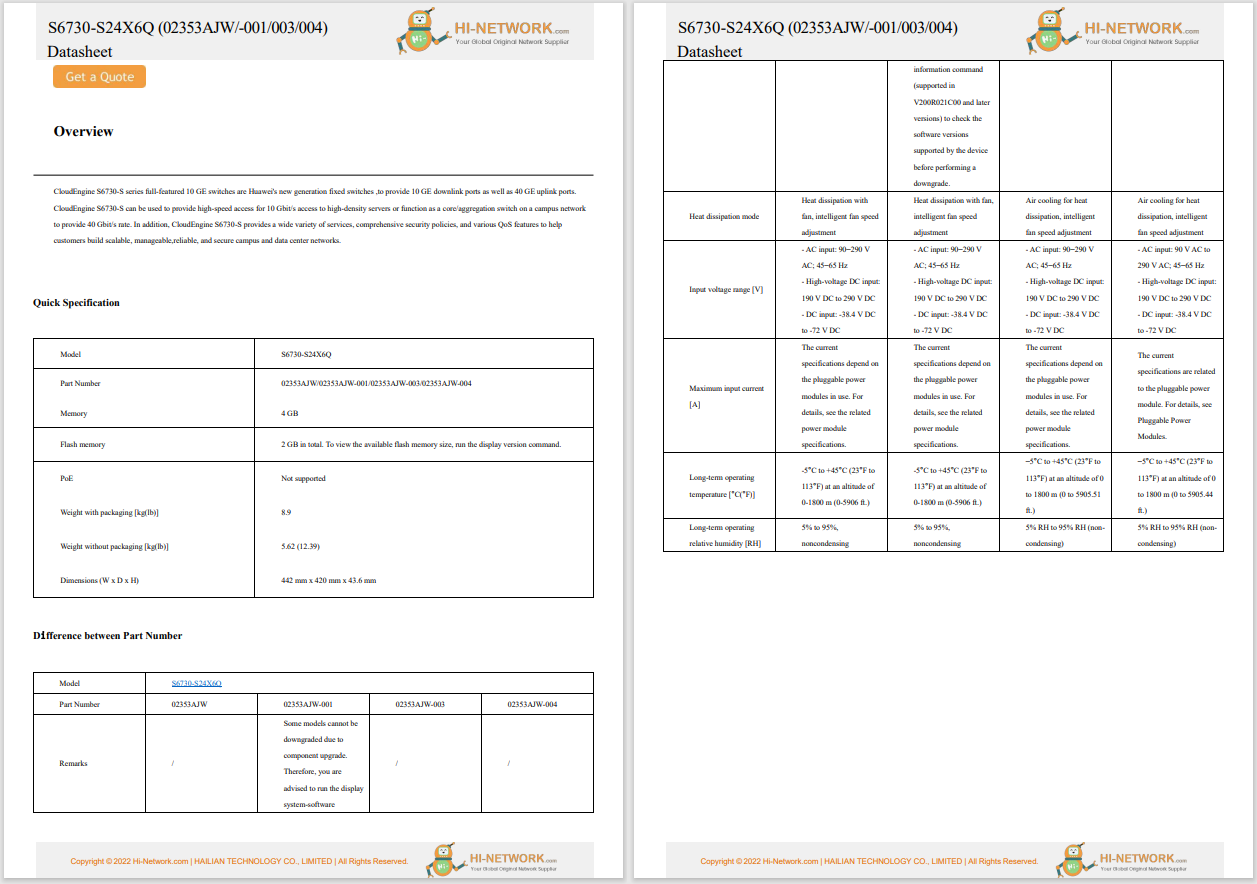





















The US Food and Drug Administration (FDA) published a guide for the public dealing with medical device cybersecurity. The guide contains three sections: (1) explanation about Internet connected medical devices and the related cybersecurity issues; (2) description of the FDA's role in regulating medical devices; (3) tips for the public on how to keep their devices safe. Among the tips provided to the public are the following: updating the devices' software, registering the device with the manufacturer, being aware if the device is not functioning appropriately, involving family or caregivers and educating them about the device, and in case of emergency to seeking medical assistance.
 Tags quentes :
Desenvolvimento de capacidades
A controvérsia do .org
Defesa do consumidor
Internet das Coisas
Tags quentes :
Desenvolvimento de capacidades
A controvérsia do .org
Defesa do consumidor
Internet das Coisas
Cadastre-se agora para Ações Semanais de Promoção
100% free, Unsubscribe any time!
Add 1: Room 605 6/F FA YUEN Commercial Building, 75-77 FA YUEN Street, Mongkok KL, HongKong Add 2: Room 405, Building E, MeiDu Building, Gong Shu District, Hangzhou City, Zhejiang Province, China
Whatsapp/Tel: +8618057156223 Tel: 0086 571 86729517 Tel em HK: 00852 66181601
Email: [email protected]
 English
English Pусский
Pусский Français
Français Español
Español Português
Português